INTACT™ Valve (Dispensing)
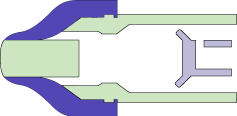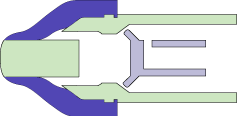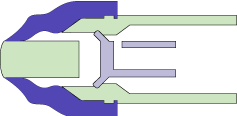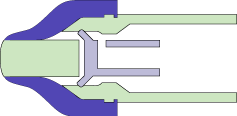
MEDInstill’s multiple-dose delivery devices utilize the patented Intact™ one-way visco-elastic valve. This valve is unique and designed to dispense product while acting as an absolute non-contamination barrier. The valve allows formulation to exit from the device while preventing ingress of air, moisture, bacteria or other contaminants that may be harmful to the product, patient or consumer.
The Intact™ valve concept is based upon the functionality of the arterial valve in the cardiovascular system. The arterial valve allows the flow of blood in only one direction using stress differential. Similarly, the Intact™ valve concept consists of:
- A tapered elastomeric valve that seals against the comparatively rigid tip of the device nozzle. This aspect is similar to the tapered walls of the arterial valve.
- Pressure from actuation due to manual or mechanical force (as compared with the pumping action of the heart).
The Intact™ valve has undergone extensive testing to assess its performance and barrier properties. These tests include FDA mandated microbiological challenge testing as well as a variety of consumer use tests. It has demonstrated the capability to prevent contamination and maintain the sterility of the contents of an Intact™ filled multiple-dose delivery device.
The Intact™ valve has been developed in a range of formats and incorporated into a wide variety of devices, containers and packages.
The Intact™ valve has been designed to handle:
- Solutions
- Gels
- Suspensions
- Creams
- Ointments
The Intact™ valve can accommodate:
- Manual operation (thumb dome, actuator, lever…)
- Electric operation (using a peristaltic pump within an automated machine for example)
Depending on the application requirements, we have several semi or fully annular valves designs to provide:
- Metered dosing
- Drops
- Streams
- Constant flows
- In 2009, the Intact™ Valve received two LACF acceptances (for low acid liquid product aseptic processing).
- The first Intact™ one-way valve was launched in Europe in June 2010.
- In January 2011, the second generation Intact™ valve received a letter of equivalence from LACF.
- In September 2011, NSF International established new requirements in NSF 18 for dispensing equipment designed to maintain the safety of potentially hazardous foods held under controlled conditions without refrigeration for dispensers that operate with a mechanical barrier that is also the original hermetic seal. As far as we know, the Intact™ technology is the only technology which has been able to meet such requirement.
- Multiple-Dose Preservative Free Sterile Dispensing
- No refrigeration needed neither before nor after opening (unless the product requires it, such as Insulin)
- Longer shelf-life
- Metered Dosing

Out-Of-Home beverages system integrating innovative Pure-Dose™ Valve technology and delivering new dispensing solution with the following technical advantages:
- Sterile non-contamination one way valve ensuring the highest food safety standards in a sector where skill levels and staff turnovers are issues
- First fully integrated, automatic espresso and cappuccino machine
- Ambient storage of milk and chocolate concentrate
- 3 major risk factors solved: improper holding temperature; contaminated equipment; poor personal hygiene
Hot and ambient beverage dispenser which delivers beverages from soluble coffee powder and aseptic liquid concentrate ingredients
- New machine concept which combines the benefits of traditional and super-automatic coffee machines
- Automatic, operator friendly machine that is easy to use and simple to clean & maintain
- High performance machine capable of dispensing premium quality beverages that one would expect from a traditional machine
- Provide consumers with a machine that offers an extended menu of hot coffee, milk and chocolate based beverages
